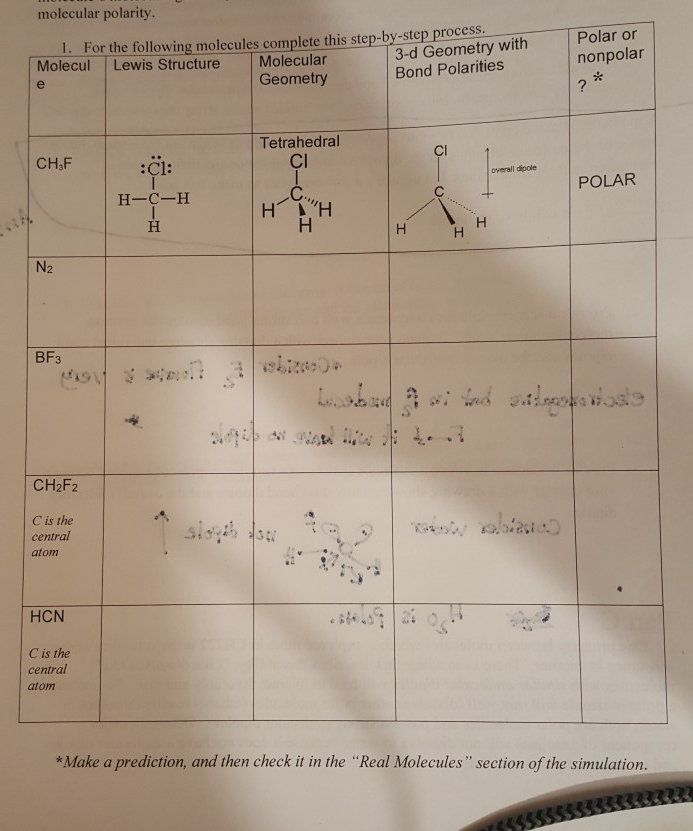
The total electrons seen in the outermost shell of any atom which has a major role in bond formation is called valence electrons. Difluoromethane, CH 2F 2 Valence electrons of bondsįormal charge of carbon, fluorine and hydrogen is zero. It can be either positive or negative and zero.įormal charge = valence electrons – no. The charge given to the atoms after bond construction is called formal charge. So total six lone pairs are present here. Out of seven outer electrons only one is used for bonding and the remaining six exist as three lone pairs. But there is lone pairs associated with two fluorine atoms. Here there is no lone pair for carbon and hydrogen. The number electrons which doesn’t take any role in bond formation is its lone pair of electrons. Shape and Angle of CH 2F 2 Difluoromethane, CH 2F 2 Lone pairs Even though hydrogen doesn’t obey octet rule it is stable. Similarly fluorine has seven before bond formation and eight after bond making.īut hydrogen needs only two electrons for stable existence and it is obtained through bonds. So it’s valence shell now filled with eight electrons. When the bond formation takes place carbon acquire four more electrons from Fluorine and hydrogen. Fluorine and hydrogen with seven and one outer electrons are present around carbon atom. 
Carbon is the middle atom and has four outer electrons. We all know that difluromethane consists of one carbon attached with two hydrogen and fluorine. There is no resonance structure found for difluromethane. It is important to retain the electronic arrangements of atoms during resonance. Usually this can be seen in double bonded compounds. Such structures are called resonance structure and process is resonance. The motion of electrons in association with an atom results more than one structure to a molecule. Lewis structure of CH2F2 Resonance in Difluoromethane, CH 2F 2 Carbon forms four steady bonds with two fluorine and hydrogen. The sum total of valence electrons in difluromethane is 20.Ĭarbon is the central atom and all the four other atoms are placed around it along with their valence electrons. So structures can be termed as Lewis dot structures.ĭifluromethane is formed by the substitution of two hydrogen atoms by fluorine. Here the electrons and bonds are denoted by dots and lines respectively. Structures of molecules drawn through this concept is an easy way for understanding the bond formation between different atoms. Lewis structure of Difluoromethane, CH 2F 2 It is used as a refrigerant and as a fire extinguisher. It’s a substance with 52.024g/mol which has the ability to undergo endothermic process. But it’s boiling point and melting point is found to be very low that is -136 0C and -52 0C respectively. It is a colorless substance with gaseous nature. It have other names like difluromethylene, HFC- 32, methylene fluoride. Two hydrogens atoms in methane is substituted by a fluorine atom. The strong carbon fluorine bonds in this molecule determines its chemical properties.ĭifluromethane is an organic dihalo compound. (CC BY-SA-NC 3.Cf2h2 lewis structure and other related facts are discussed in this article.ĭifluoromethane is a haloalkane compound with two fluorine atoms. Hence the vector sum is not zero, and H 2O has a net dipole moment. (b) In H 2O, the O–H bond dipoles are also equal in magnitude, but they are oriented at 104.5° to each other. Their vector sum is zero, so CO 2 therefore has no net dipole. (a) In CO 2, the C–O bond dipoles are equal in magnitude but oriented in opposite directions (at 180°). \): How Individual Bond Dipole Moments Are Added Together to Give an Overall Molecular Dipole Moment for Two Triatomic Molecules with Different Structures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
